Research
Host-pathogen co-adaptation during chronic infections
Chronic bacterial infections affect millions of people worldwide and are often resistant to antibiotic treatment. During these infections, pathogenic bacteria establish long-term interactions with host tissues, leading to co-adaptation. This process not only allows bacteria to adapt to their new environment but can also induce host tissue adaptations that influence health and disease progression. Our goal is to understand these co-adaptations and leverage this knowledge to improve treatment efficacy.
We are particularly focused on chronic lung infections. To investigate how bacterial pathogens grow, survive, and interact with lung cells, we are building an interdisciplinary team that will include microbiologists, tissue engineers, developmental biologists, immunologists, and computational biologists. By using advanced in vitro tissue models (tissue culturing and organoids), combined with clinical data and patient samples, we examine the physiological dynamics of both pathogen and host during these infections.
ongoing projects

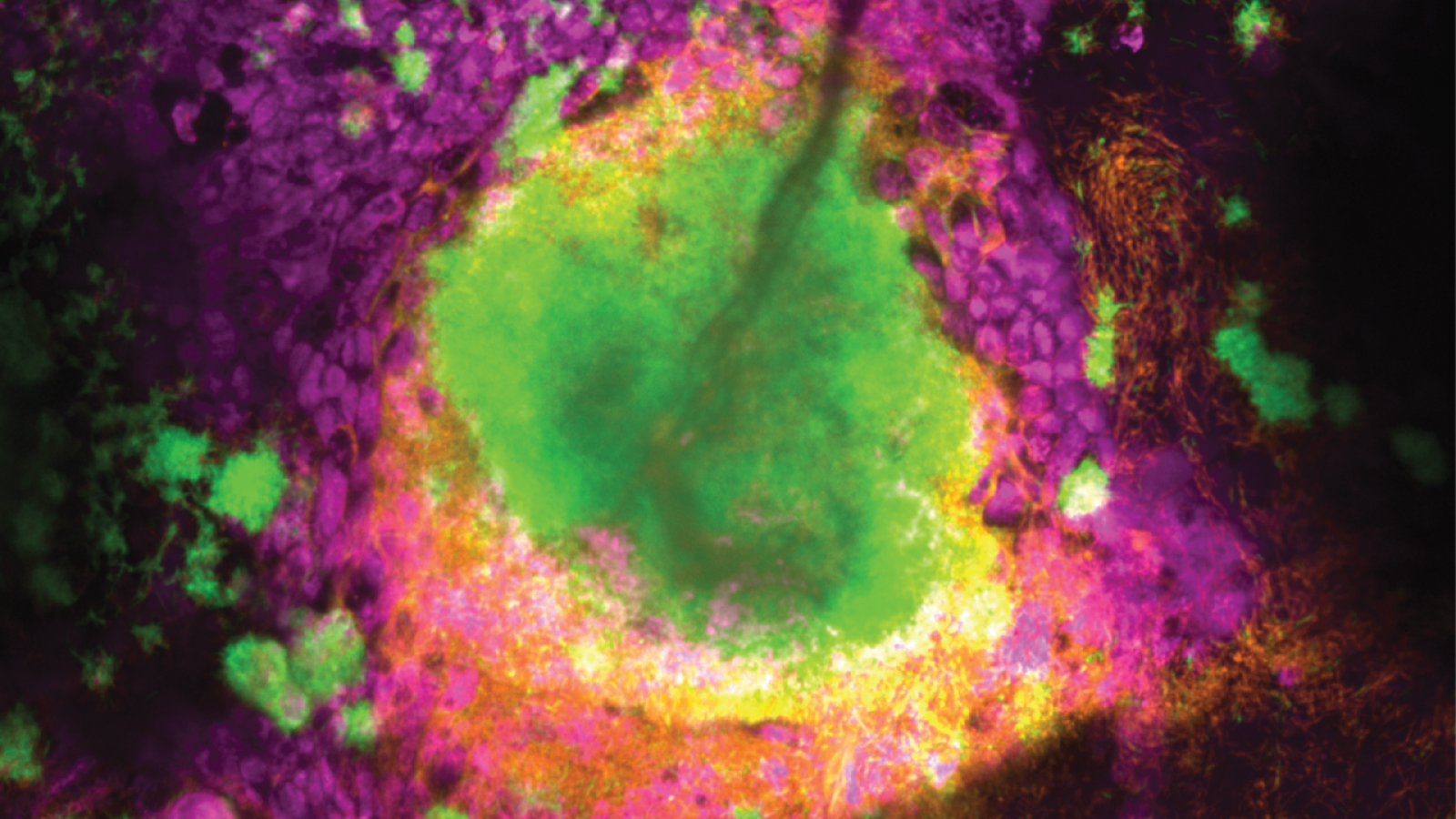
Pathogen life at the airway mucosal surface
We have developed a roadmap that combines tissue culturing, high-throughput omics assays, and live imaging to study how bacterial pathogens colonize the mucosal surface and interact with the airway epithelium at the single-cell level (see this and this). Our focus is on understanding the mechanisms that drive pathogen growth, tissue damage, and antibiotic resistance. We are interested in hard-to-treat pathogens such as Pseudomonas aeruginosa and Nontuberculous mycobacteria (NTMs) as well as highly prevalent bacteria such as the human-adapted pathogen Non-typeable Haemophilus influenzae (NTHi). Together, these pathogens contribute significantly to chronic lung diseases such as chronic obstructive pulmonary disease (COPD), bronchiectasis, and cystic fibrosis (CF).

Understanding how pathogens modulate tissue regeneration
In addition to studying pathogen behavior, we are exploring how the airway epithelium adapts to chronic infections. Bacterial colonization triggers inflammation and tissue damage, contributing to disease progression. While bacterial-induced airway remodeling plays a role in chronic lung disease, the underlying mechanisms are not yet fully understood. We use our in vitro tissue models to investigate how pathogens influence stem cell behavior and interfere with tissue regeneration. By integrating this approach with clinical data and patient samples, we aim to uncover how microbes impact disease progression and tissue repair.

